Characteristics of Insect Cell Baculovirus Expression Vector System
2021-12-03
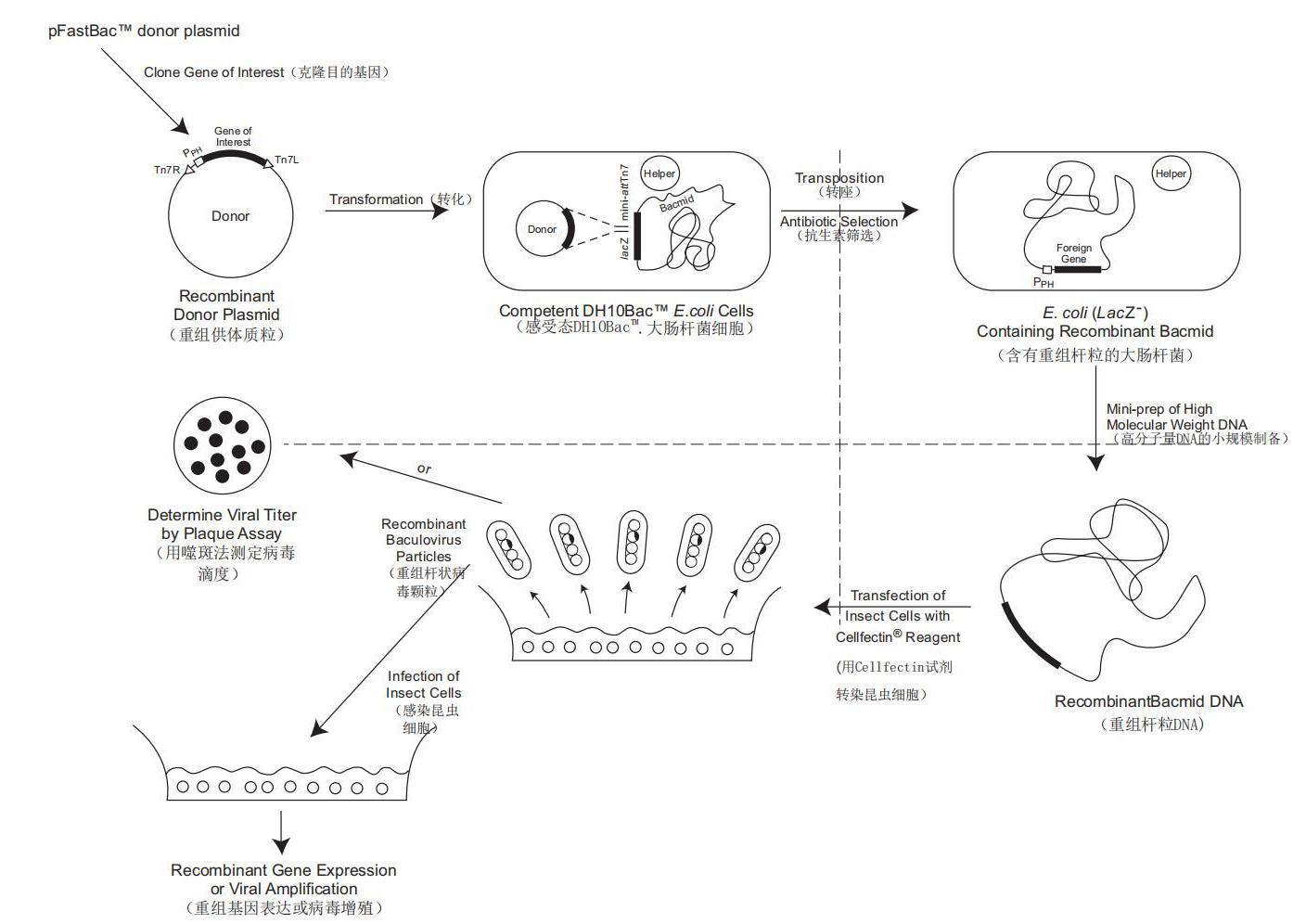
Insect cell baculovirus expression vector system (IC-BEVS) is one of the four major expression systems (insect cell, bacterial, yeast and mammalian cell expression systems).With various advantages, IC-BEVS is an important source for the development of new drugs, materials and reagents, widely used for the study of recombinant protein expression[1], production of human and veterinary vaccines and for pharmaceutical application. For example, both Cervarix, GlaxoSmithKline's cervical cancer vaccine and Flubolk, Sanofi's tetravalent influenza vaccine adopt IC-BEVS.The recombinant COVID-19 vaccine (Sf9 cell), jointly developed by WestVac Biopharma Co., Ltd. and the State/National Key Lab of Biotherapy, West China Hospital of Sichuan University, is also based on IC-BEVS, where the genes of SARS-CoV-2 are introduced into insect cells multiplied in the culture solution to prepare the SARS-CoV-2 S protein, inducing the human body to produce antibodies to block the viral infection. In addition, for the new generation vaccine against Delta and Omicron variants, WestVac has precisely designed the subunit vaccine antigen with stable self-assembled trimeric sequence, on the mature IC-BEVS platform. The vaccine has shown excellent immunogenicity and safety.Insect Cell Baculovirus Expression Vector System, IC-BEVS
Practices indicate that IC-BEVS is a highly versatile system able to express and produce one recombinant protein or express multiple genes simultaneously. It is widely applied in a variety of studies for the following advantages [2]:1. High expression of the target geneBaculvirus contains POLH, which is used to produce target protein in a large amount within short time.2. High versatilitySingle cell line-baculovirus backbone may express any gene, and modification to the standard gene expression process can be made to optimize particular protein production. IC-BEVS also enables multiple larger genes to be embedded into the baculovirus genome and express multiple proteins. Over 500 functional recombinant proteins have been successfully expressed on IC-BEVS.3. High speedThe construction of recombinant baculovirus is fully standardized and may be used to produce and obtain large quantities of recombinant protein at a higher speed.4. Post-translational modification [3]Insect cell lines may perform post-translational modification of most eukaryotic-derived recombinant proteins. Some proteins that are impossible or difficult to be expressed or modified in other systems can be easily expressed in IC-BEVS while maintaining the biological activity.5. Scale-upInsect cells may be cultured in suspension, hence can be cultured in some large fermentation tanks for rapid production of biologics.6. SafetyThe process of protein production with IC-BEVS is classified as BSL-1 which is relatively safe for experimental studies and production.References:1.Hitchman Richard B,Possee Robert D,King Linda A. High-throughput baculovirus expression in insect cells.[J]. Methods in Molecular Biology,2011,824. https://doi.org/10.1007/978-1-61779-433-9_33.2. Hofmann C, Sandig V, Jennings G, etal. Efficient gene transferinto human hepatocytes by baculovirus vectors[J]. Proc Natl Acad Sci USA, 1995(92): 10099-10103.3.King AMQ, Adams MJ, Carstens EB, et al. Ninth Report of the International Committee on Taxonomy of Viruses. London: Elsevier, 2012